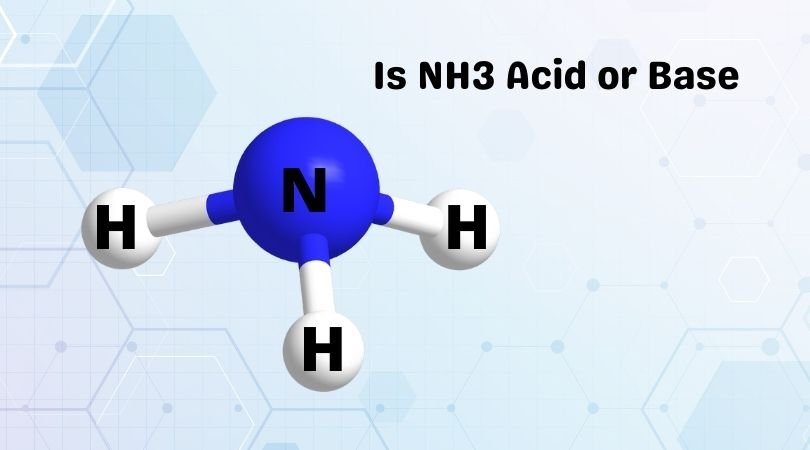
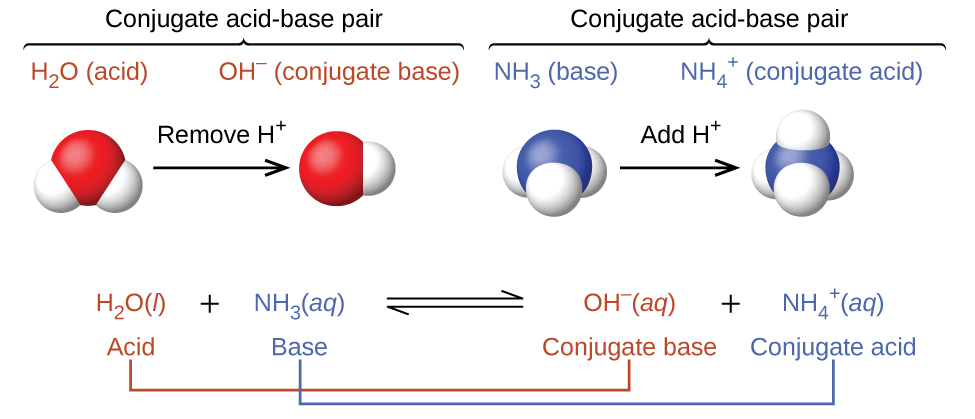
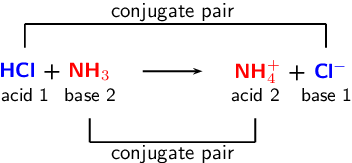
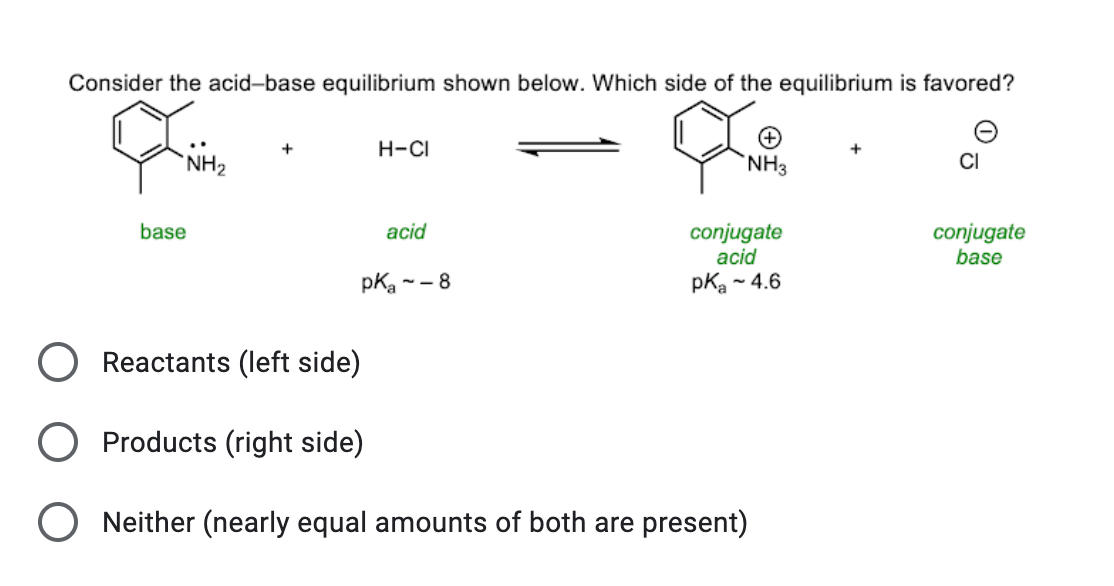
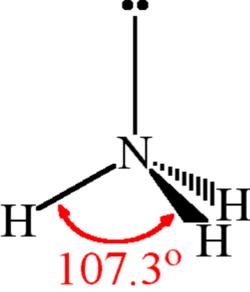
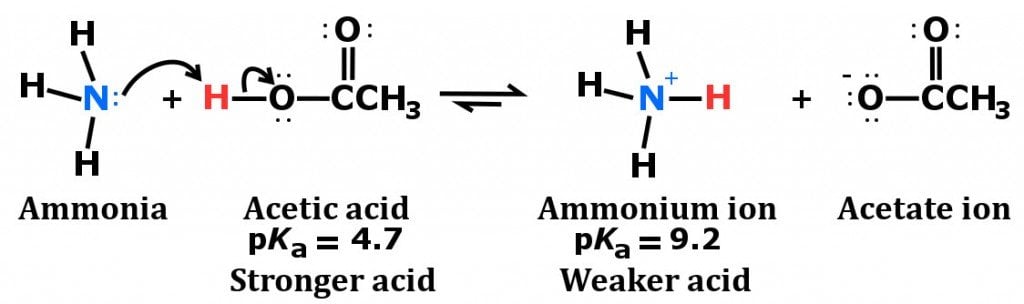
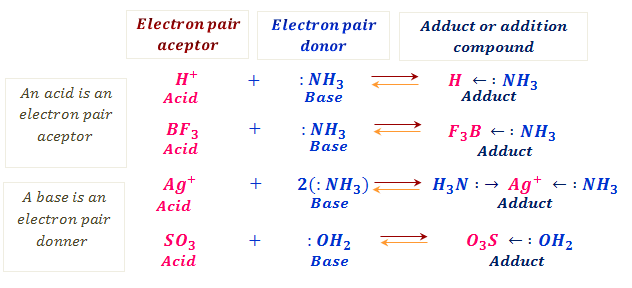
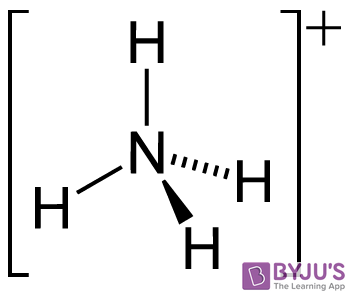
Spontaneity of the acid–base reaction between acetic acid and ammonia... | Download Scientific Diagram

SOLVED: Identify the conjugate acid-base pairs in the reaction shown below HBr + NH3 Br + NH4 acid; chemPad Help Gianke conjugate base: chemPad Help Girekt base: chemPad Help Creke conjugate acid:

Identify the conjugate acid-base pairs in this equilibrium. NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com

Electric Field-Driven Acid−Base Chemistry: Proton Transfer from Acid (HCl) to Base (NH3/H2O) | The Journal of Physical Chemistry A
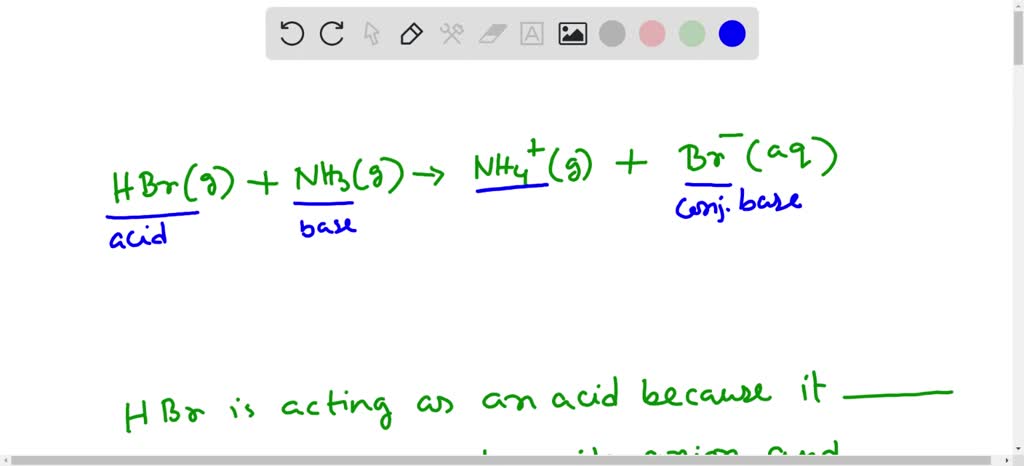
SOLVED: In the following acid-base reaction: HBr(g) + NH3(g) → NH4+(aq) + Br– (aq) HBr is acting as the acid, because it a proton to form bromide anion, and NH3 is acting