
Natriumphosphat, dibasisches Dihydrat, entspricht den analytischen Spezifikationen von BP, Ph. Eur., 98.5 bis 101 % (Berechnung basiert auf trockener Substanz), Honeywell Fluka | Fisher Scientific

SOLVED: What is the balanced equation and the net ionic equation. NaH2PO4 + Na2HPO4 + H2O + MgCl2 ⇒ NaH2PO4 + Na2HPO4 + H2O + NaCl ⇒ ( we did an experiment

Equilibrium constants is given (in atm) for the following reaction 0^∘C : Na2HPO4. 12H2O(s) Na2HPO4. 7H2O(s) + 5H2O(g) ; Kp = 2.43 × 10^-13 The vapour pressure of water at 0^∘C is

1 Bán Hóa chất di-Sodium hydrogen phosphate dihydrate, - Na2HPO4.2H2O - SO0339 - Scharlau giá rẻ ở hcm

Comparative Study of Sodium Phosphate and Sodium Sulfate in Aqueous Solutions at (298.15 to 353.15) K | Journal of Chemical & Engineering Data

Measurement and Modeling of the Solubility of NH4VO3 in the Na2HPO4–H2O and (NH4)2HPO4–H2O Systems | Journal of Chemical & Engineering Data




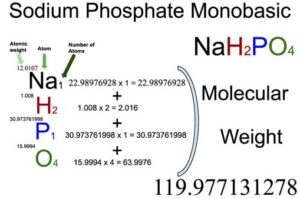


![Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/01/sodium-phosphate-dibasic-dihydrate-molecular-weight-calculation-300x186.jpg)

2MoO4%20+%20HNO3%20=%20(NH4)3(PMo12O40)%20+%20NH4NO3%20+%20NaNO3%20+%20H2O.svg)