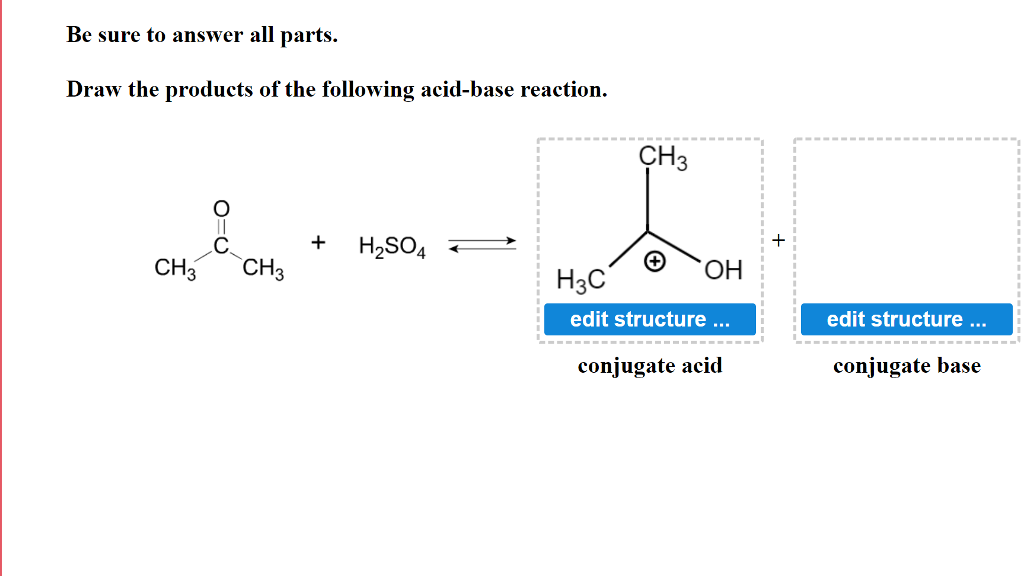
✓ Solved: Hydrazine, N2H4, is a weak base and can react with an acid such as sulfuric acid: 2N2H4(aq)+H2SO4(aq)→...

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa