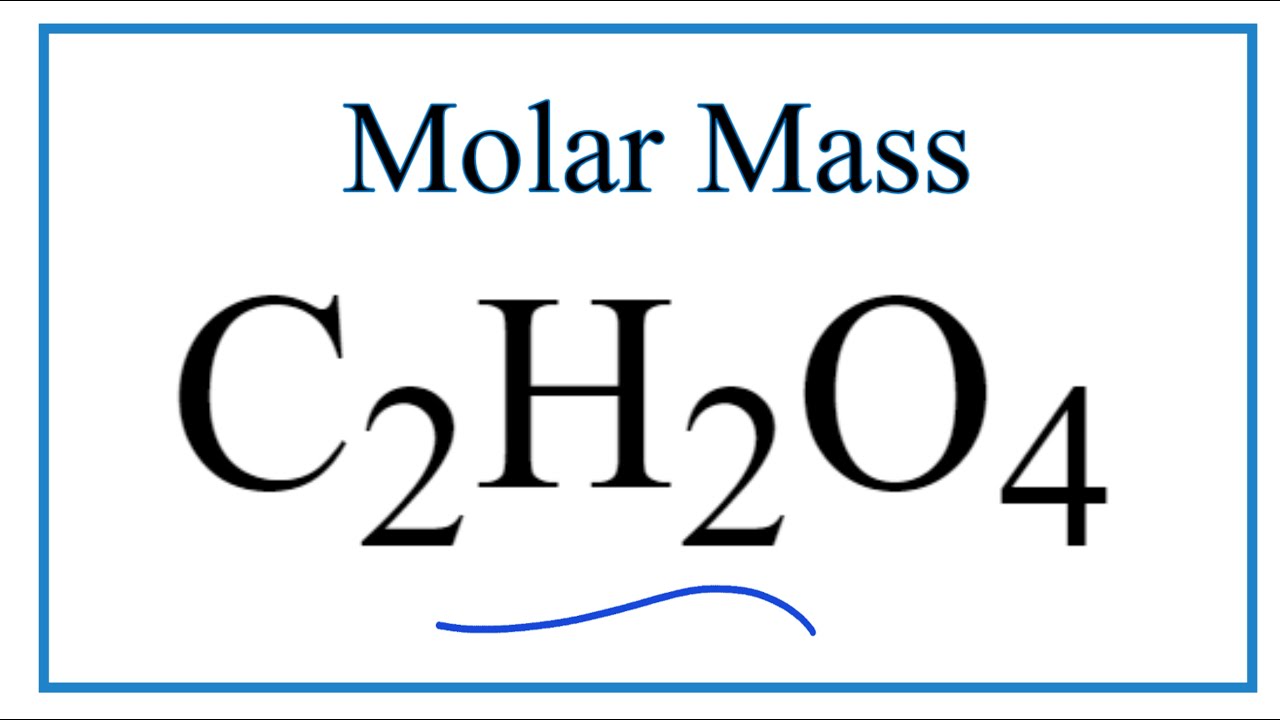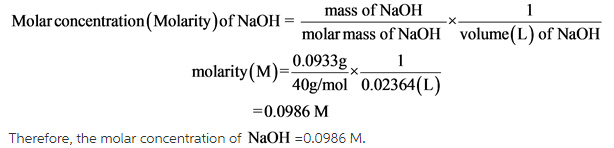
Oxalic acid, H2C2O4*2H2O (molar mass = 126.07 g/mol) is oftenused as a primary standard - Home Work Help - Learn CBSE Forum

Weight of H2C2O4 2H2O required to prepare 500ml of 0 2N solution is - Chemistry - Solutions - 12713077 | Meritnation.com

What mass of oxalic acid dihydate, H2C2O4•2H2O, is needed to make a 0.498 M solution of oxalic acid in a 250.0 mL volumetric flask? - Quora






![Solved 3. In the preparation of cis-K[Cr(C2O4)2(H20)2] 2H2O, | Chegg.com Solved 3. In the preparation of cis-K[Cr(C2O4)2(H20)2] 2H2O, | Chegg.com](https://media.cheggcdn.com/study/532/5329fd26-00bf-4ded-ba47-a0acbb56126a/image.png)

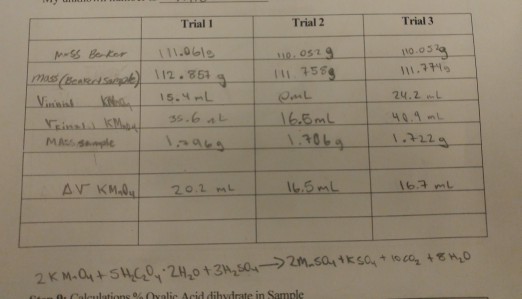

![Oxalic Acid Dihydrate [H2C2O4 2H2O ] [CAS_6153-56-6] 99.6+% Fine White – Wintersun Oxalic Acid Dihydrate [H2C2O4 2H2O ] [CAS_6153-56-6] 99.6+% Fine White – Wintersun](https://cdn.shopify.com/s/files/1/0724/7981/products/15-005-4_1024x1024.jpg?v=1662152063)