Introduction to Regulatory Affairs: 3. Roles of the European Directorate for the Quality of Medicines (EDQM) and the European Pharmacopoeia (EP) Commission in the medicines regulations system
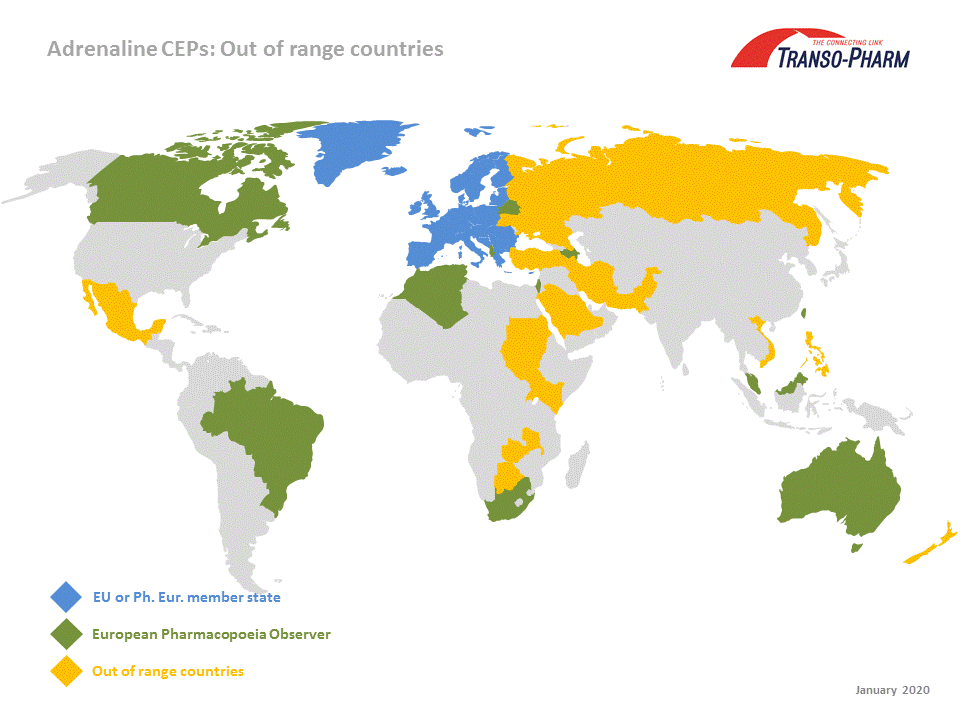
EDQM reminds CEP holders of their responsibilities towards their customers - European Directorate for the Quality of Medicines & HealthCare
CEP of the future”: second project update - European Directorate for the Quality of Medicines & HealthCare



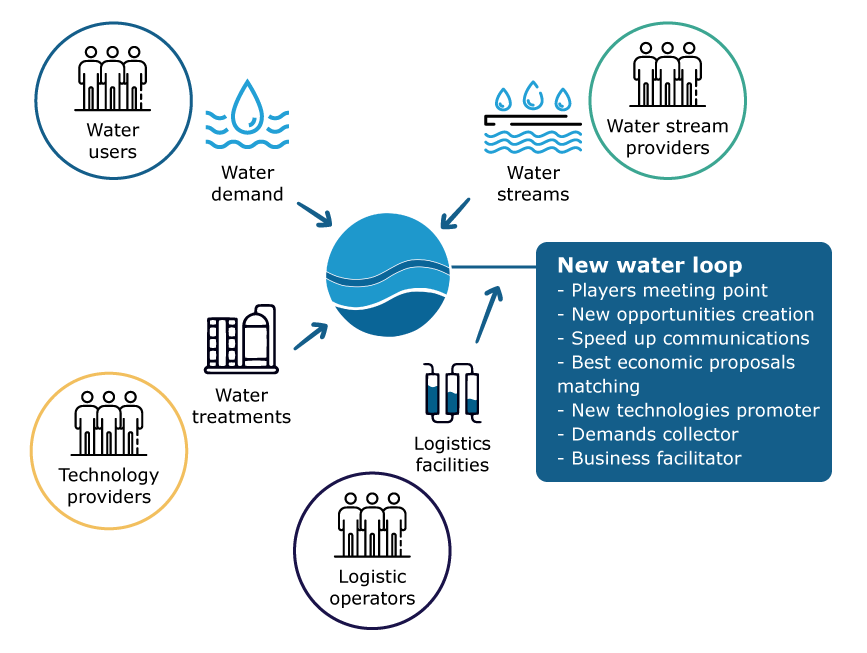




.jpg)