![Ni(II) complexes with thioether-functionalized silylamide ligands. Synthesis and crystal structures of [Ni{Me2Si(N-C6H4-2-S-t-Bu)2}], [Ni{Ph2Si(N-C6H4-2-SMe)2}] and [Ni{Ph2Si(N-C6H4-2-SPh)2}] Ni(II) complexes with thioether-functionalized silylamide ligands. Synthesis and crystal structures of [Ni{Me2Si(N-C6H4-2-S-t-Bu)2}], [Ni{Ph2Si(N-C6H4-2-SMe)2}] and [Ni{Ph2Si(N-C6H4-2-SPh)2}]](https://www.degruyter.com/document/doi/10.1515/znb-2018-0263/asset/graphic/j_znb-2018-0263_scheme_001.jpg)
Ni(II) complexes with thioether-functionalized silylamide ligands. Synthesis and crystal structures of [Ni{Me2Si(N-C6H4-2-S-t-Bu)2}], [Ni{Ph2Si(N-C6H4-2-SMe)2}] and [Ni{Ph2Si(N-C6H4-2-SPh)2}]
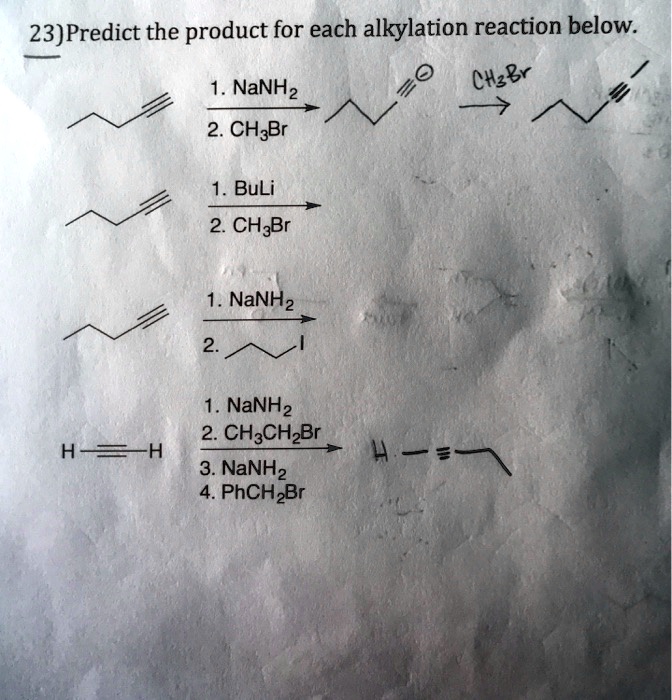
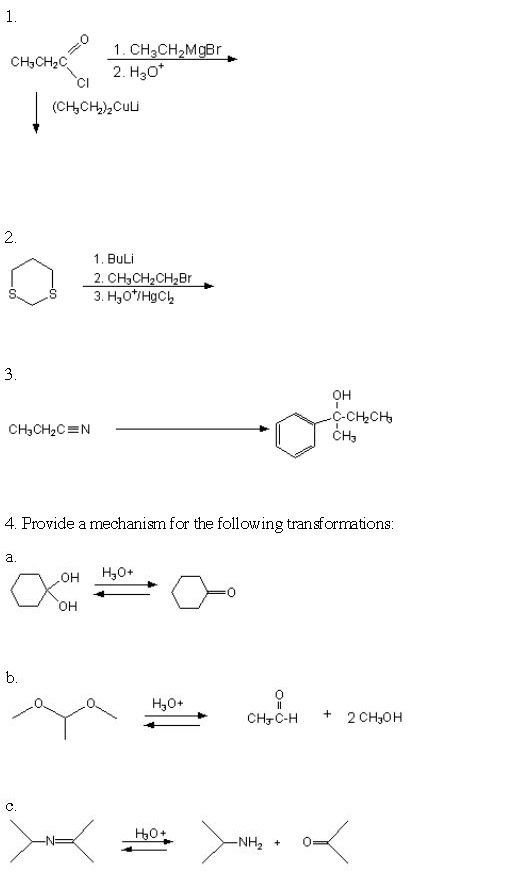
SOLVED: 23)Predict the product for each alkylation reaction below: 0 NaNH2 Chgbr 2. CHBr BuLi 2. CHzBr NaNHz NaNHz 2. CH3CHzBr 3. NaNHz PhCHzBr HS=H

Crystal Structures of n-BuLi Adducts with (R,R)-TMCDA and the Consequences for the Deprotonation of Benzene | Journal of the American Chemical Society

NMR and DFT Studies with a Doubly Labelled 15N/6Li S‐Trifluoromethyl Sulfoximine Reveal Why a Directed ortho‐Lithiation Requires an Excess of n‐ BuLi - Hédouin - Angewandte Chemie International Edition - Wiley Online Library


![Molbank | Free Full-Text | Benzo[b]thiophene-2-carbaldehyde Molbank | Free Full-Text | Benzo[b]thiophene-2-carbaldehyde](https://www.mdpi.com/molbank/molbank-2014-M823/article_deploy/html/images/molbank-2014-M823-sch002.png)